Combustion and Flame Class 8 Science : Comprehensive study resource for CBSE Science Chapter 6 “Combustion and Flame Class 8”. Includes detailed revision notes, important subjective questions and answers, and MCQs for exam preparation.
Table of Contents
📘 SECTION 1: DETAILED REVISION NOTES – Combustion and Flame Class 8
# Combustion and Flame Class 8
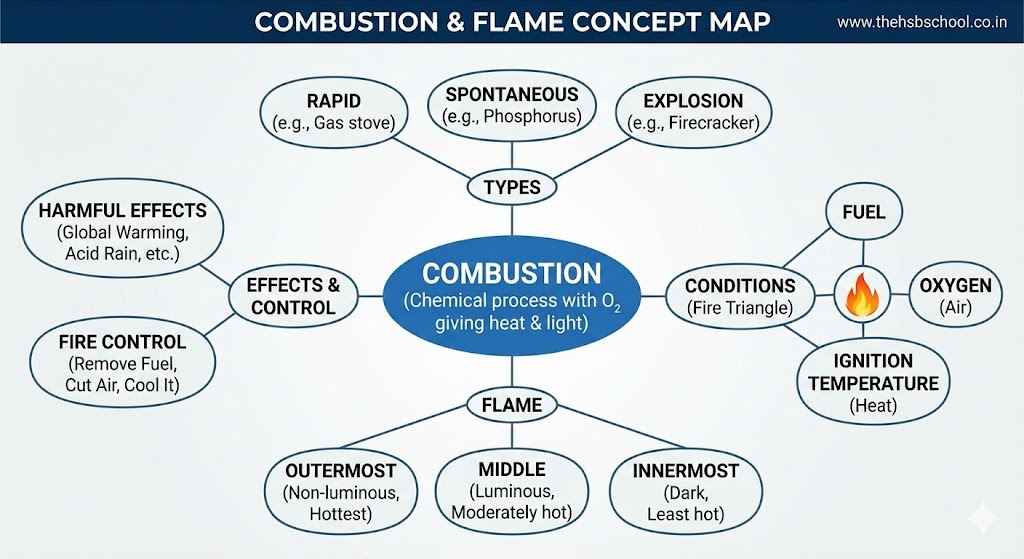
1. What is Combustion?
- Definition: A chemical process in which a substance reacts with oxygen to give off heat is called combustion. Sometimes, light is also given off during combustion, either as a flame or as a glow.
- Combustible Substances (Fuels): The substances which undergo combustion are called combustible substances. Examples: Wood, coal, charcoal, petrol, diesel, LPG (Liquefied Petroleum Gas), CNG (Compressed Natural Gas), magnesium ribbon.
- Non-Combustible Substances: Substances that do not burn in air. Examples: Stone, glass, iron nails, water, sand.
2. Conditions Necessary for Combustion
For combustion to take place, three conditions are essential (often called the “Fire Triangle”):
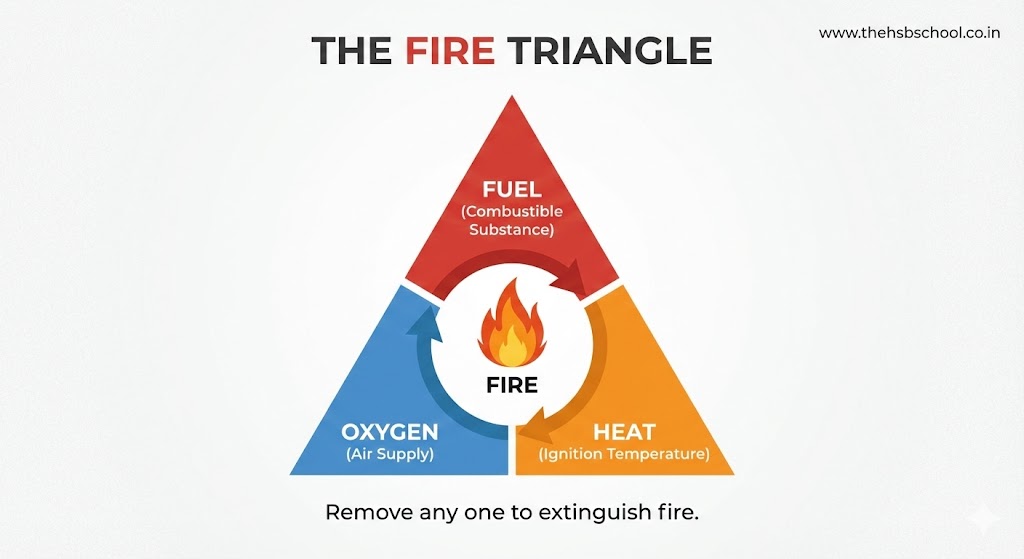
- Presence of a Combustible Substance (Fuel): Something that can burn.
- Presence of Air (Oxygen): Oxygen is a supporter of combustion. Without it, fire cannot be sustained.
- Attainment of Ignition Temperature:
- Ignition Temperature: The lowest temperature at which a substance catches fire and starts burning.
- A substance will not catch fire if its temperature is lower than its ignition temperature. (Example: Paper by itself burns easily, but paper wrapped around an aluminium pipe filled with water does not burn easily because the water absorbs the heat, preventing the paper from reaching its ignition temperature).
- Inflammable Substances: Substances that have very low ignition temperatures and can easily catch fire with a flame are called inflammable substances (e.g., petrol, alcohol, LPG).
3. How Do We Control Fire?
To extinguish a fire, we must remove at least one of the three conditions necessary for combustion:
- Removing the fuel: (Often difficult in large fires).
- Cutting off the air supply: Using blankets, sand, or carbon dioxide.
- Bringing down the temperature below the ignition temperature: Using water.
Types of Fire Extinguishers:
- Water: Works best for wood or paper fires. It cools the combustible material and produces water vapour which cuts off the air supply.
- Caution: Never use water on electrical fires (water conducts electricity) or oil/petrol fires (oil floats on water and keeps burning).
- Carbon Dioxide (CO₂): The best extinguisher for electrical equipment and inflammable liquids like petrol.
- CO₂ is heavier than oxygen and covers the fire like a blanket, cutting off oxygen contact.
- In cylinders, CO₂ is stored as a liquid under high pressure. When released, it expands enormously and cools down, lowering the temperature of the fuel.
- Sand/Soil: Useful for small oil fires to cut off the air supply.
4. Types of Combustion
- Rapid Combustion: When a substance burns rapidly and produces heat and light. External heat is usually required to start it. (e.g., burning of LPG in a gas stove, burning of a matchstick).
- Spontaneous Combustion: When a material suddenly bursts into flames without the application of any apparent cause (no external heating required). (e.g., Phosphorus burns in air at room temperature; forest fires caused by the heat of the sun or lightning).
- Explosion: A sudden reaction that occurs with the evolution of heat, light, and sound. A large amount of gas is liberated in the reaction. (e.g., bursting of firecrackers).
5. Flame
- Definition: A flame is a region where the combustion of gaseous substances takes place.
- Substances that vaporize during burning produce flames (e.g., wax candle, kerosene oil). Substances that do not vaporize (e.g., charcoal) do not produce a flame; they just glow.
Structure of a Candle Flame: A candle flame has three main zones:
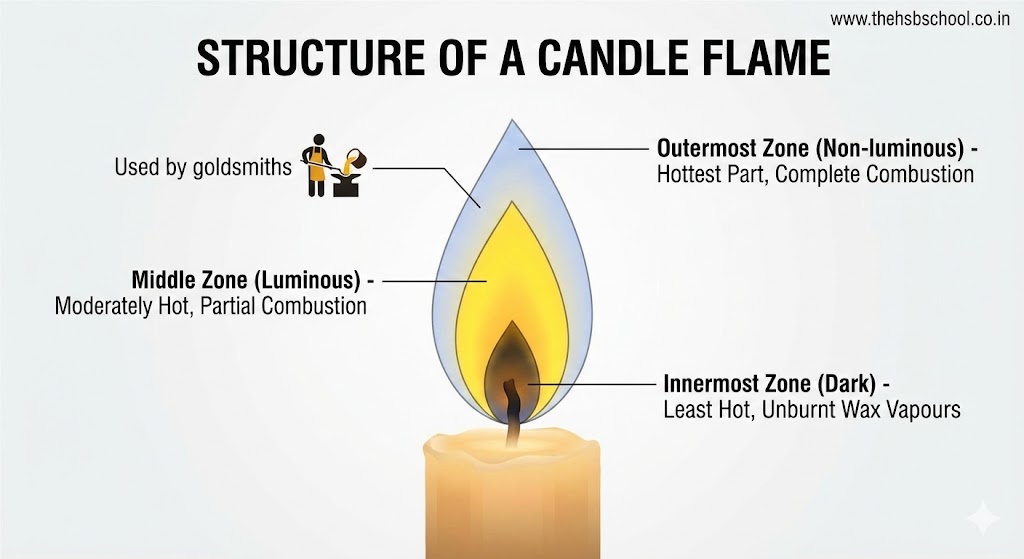
- Innermost Zone (Dark Zone):
- Area surrounding the wick.
- Contains unburnt vapours of wax.
- It is the ‘least hot’ part of the flame.
- Black in colour due to unburnt carbon particles.
- Middle Zone (Luminous Zone):
- The bright yellow part of the flame.
- Partial (incomplete) combustion takes place here due to limited oxygen supply.
- It is moderately hot.
- Produces light and soot (unburnt carbon particles).
- Outermost Zone (Non-luminous Zone):
- The faintly visible blue part at the edges.
- Complete combustion takes place here due to ample oxygen supply.
- It is the ‘hottest’ part of the flame.
- Goldsmiths use this outermost zone to melt gold and silver because it is the hottest region.
6. Fuel and Fuel Efficiency
- Fuel: Any substance that is burnt to produce heat energy for domestic and industrial purposes.
- Ideal Fuel: An ideal fuel should be cheap, easily available, burn easily in air at a moderate rate, produce a large amount of heat, and leave behind no undesirable residue. (No fuel is perfectly “ideal,” LPG is considered a very good fuel).
- Fuel Efficiency (Calorific Value):
- The amount of heat energy produced on complete combustion of 1 kg of a fuel is called its calorific value.
- The unit of calorific value is kilojoule per kilogram (kJ/kg).
- Higher calorific value means the fuel is more efficient. (e.g., Hydrogen has the highest calorific value, much higher than wood or coal).
7. Harmful Effects of Burning Fuels
Increasing fuel consumption has harmful effects on the environment:
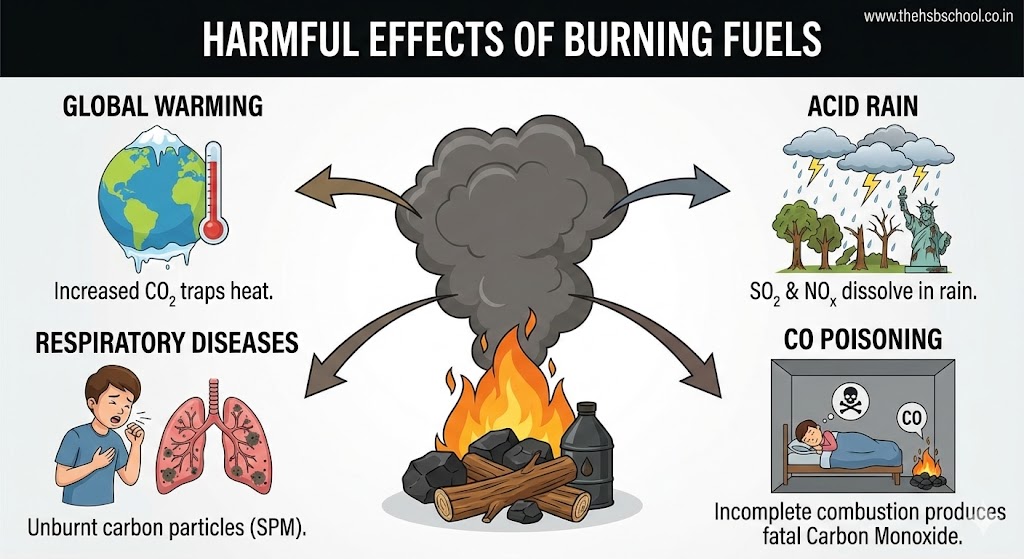
- Respiratory Diseases: Burning of carbon fuels like wood, coal, and petroleum releases unburnt carbon particles (particulate matter). These fine particles are dangerous pollutants causing respiratory diseases like asthma.
- Carbon Monoxide Poisoning: Incomplete combustion of fuels gives carbon monoxide (CO) gas. It is a very poisonous gas. It is dangerous to burn coal in a closed room because the CO produced can kill persons sleeping in that room.
- Global Warming: Combustion of most fuels releases carbon dioxide (CO₂). Increased concentration of CO₂ in the air is a major cause of global warming (rise in Earth’s temperature), leading to melting polar glaciers and rising sea levels.
- Acid Rain: Burning of coal and diesel releases Sulphur dioxide (SO₂) gas. Petrol engines release oxides of Nitrogen. These gases dissolve in rain water and form acids. Such rain is called acid rain. It is very harmful to crops, buildings (like the Taj Mahal), and soil.
🗣️ SECTION 2: SUBJECTIVE QUESTION AND ANSWERS – Combustion and Flame Class 8
Q1. Define ignition temperature. Ans: The minimum temperature at which a substance catches fire and starts burning is called its ignition temperature.
Q2. State the three essential conditions required for combustion to take place. Ans: The three essential conditions are:
- Presence of a combustible substance (fuel).
- Presence of a supporter of combustion (air/oxygen).
- Heating the substance to its ignition temperature.
Q3. Why is water not used to control fires involving electrical equipment? Ans: Ordinary water may contain salts and conduct electricity. If used on an electrical fire, the person handling the water hose might get an electric shock, which can be fatal.
Q4. Why does a goldsmith use the outermost zone of the flame for melting gold and silver? Ans: The outermost zone of the flame undergoes complete combustion and is the hottest part of the flame. Goldsmiths need a very high temperature to melt metals like gold and silver, so they use this zone.
Q5. Explain how CO₂ is able to control fires. Ans: Carbon dioxide (CO₂) is heavier than oxygen. It covers the fire like a blanket. Since the contact between the fuel and oxygen is cut off, the fire is controlled. Also, when released from cylinders, CO₂ expands rapidly and cools down, lowering the temperature of the fuel below its ignition temperature.
Q6. Differentiate between Rapid Combustion and Spontaneous Combustion. Ans:
- Rapid Combustion: Combustion that occurs rapidly, producing heat and light, usually requiring an external heat source to start (e.g., lighting a gas stove).
- Spontaneous Combustion: Combustion in which a material suddenly bursts into flames without the application of any apparent cause or external heating (e.g., phosphorus burning at room temperature).
Q7. Give reasons: Paper by itself catches fire easily whereas a piece of paper wrapped around an aluminium pipe does not. Ans: Paper has a low ignition temperature and catches fire easily. However, when wrapped around an aluminium pipe, the heat supplied to the paper is transferred to the aluminium pipe by conduction. Aluminium being a good conductor absorbs the heat, preventing the paper from reaching its ignition temperature. Hence, it does not burn easily.
Q8. Explain the different zones of a candle flame. Ans: A candle flame has three zones:
- Innermost Zone (Dark): Contains unburnt wax vapours. It is the least hot region.
- Middle Zone (Luminous/Yellow): Here partial combustion takes place due to limited oxygen. It emits yellow light and is moderately hot. It produces soot.
- Outermost Zone (Non-luminous/Blue): Here complete combustion takes place due to sufficient oxygen. It is the hottest zone and emits faint blue light.
Q9. Define Calorific Value. What is its unit? Ans: The calorific value of a fuel is the amount of heat energy produced on complete combustion of 1 kg of that fuel. Its unit is kilojoule per kilogram (kJ/kg).
Q10. How does burning of fossil fuels cause acid rain? What are its harmful effects? Ans: Burning fossil fuels like coal and diesel releases sulphur dioxide (SO₂), and petrol engines release oxides of nitrogen. These gases dissolve in rainwater to form sulphuric acid and nitric acid, respectively. This rain falls as acid rain. Harmful effects: It damages buildings and historical monuments (marble cancer), makes soil acidic affecting agriculture, and destroys aquatic life.
✅ SECTION 3: MULTIPLE CHOICE QUESTIONS (MCQs) – Combustion and Flame Class 8
1. The chemical process in which a substance reacts with oxygen to give off heat is called: a) Conduction b) Combustion c) Radiation d) Explosion
Correct Answer: b) Combustion
2. The lowest temperature at which a substance catches fire is called its: a) Boiling point b) Melting point c) Ignition temperature d) Critical temperature
Correct Answer: c) Ignition temperature
3. Which of the following is NOT a combustible substance? a) Wood b) Paper c) Iron nail d) Kerosene
Correct Answer: c) Iron nail
4. The hottest zone of a candle flame is: a) The innermost zone b) The middle zone c) The outermost zone d) The bottom zone
Correct Answer: c) The outermost zone
5. Which gas is a major cause of global warming? a) Nitrogen b) Oxygen c) Carbon dioxide d) Carbon monoxide
Correct Answer: c) Carbon dioxide
6. The unit of calorific value of a fuel is: a) kg/kJ b) kJ/kg c) Joule/kg d) Watt
Correct Answer: b) kJ/kg
7. Incomplete combustion of a fuel gives: a) Carbon dioxide b) Carbon monoxide c) Nitrogen oxide d) Sulphur dioxide
Correct Answer: b) Carbon monoxide
8. The best fire extinguisher for fires involving electrical equipment and inflammable materials like petrol is: a) Water b) Sand c) Carbon dioxide d) Foam
Correct Answer: c) Carbon dioxide
9. Phosphorus burns in air at room temperature. This is an example of: a) Rapid combustion b) Spontaneous combustion c) Explosion d) Slow combustion
Correct Answer: b) Spontaneous combustion
10. Acid rain is caused by the oxides of: a) Carbon and Nitrogen b) Sulphur and Nitrogen c) Carbon and Sulphur d) Phosphorus and Sulphur
Correct Answer: b) Sulphur and Nitrogen
11. Substances which vaporize during burning give: a) Sound b) Flame c) Smoke only d) None of these
Correct Answer: b) Flame
12. Which part of the flame does a goldsmith use for melting gold and silver? a) Dark Zone b) Luminous Zone c) Non-luminous zone d) Middle zone
Correct Answer: c) Non-luminous zone
#Combustion and Flame Class 8
